In order to deepen our knowledge of Parkinson's disease, in 2016 we set up a prospective national cohort gathering all patients with this pathology or a related syndrome followed in the network's centers. This cohort has no equivalent in Europe and internationally and will enable future industrial and academic collaborations. Its success is based on a large-scale collaborative effort and a qualitative commitment, thanks to which we can provide the scientific community with a homogeneous longitudinal data set that can be used to answer scientific questions.
Objective of the cohort
The challenge is twofold:
- to better manage patients medically by developing predictive and personalized medicine,
- to promote the development of precision therapies by targeting homogenous populations in terms of the involved pathophysiological mechanisms.
The success of the NS-PARK cohort is based on a large-scake collaborative effort and a qualitative commitment, thanks to which the NS-PARK/F-CRIN network can provide a homogenous longitudinal data set to the scientific community that can be used to solve scientific questions.
Objective of the cohort: The analysis of the collected data, clinical but also biological and imaging, will make it possible to describe the natural history of Parkinson's disease, its progression and the response to treatment.
Regulatory framework: the NS-PARK cohort received the agreement from the Comité de Protection des Personnes Ile de France 1 (Ethics committee) on 19/06/2020 and the autorization of the CNIL (French data protection authority) on 12/02/2021.
Definitions:
Cohorts : cohorts are composed of a group of people who are individually followed over time. Their purpose is to identify the occurrence of health events of interest and the related risk or protective factors. Cohorts are one of the reference instrument for epidemiological and public health research.
https://www.inserm.fr/en/our-research/public-health-research/
Longitudinal study: study that observes a phenomenon or a population repeatedly over time to analyze the evolution of an event (as opposed to a cross-sectional study which focuses on a specific moment in time).
Participation in the cohort
Inclusion
Inclusion in the cohort is proposed to all patients with Parkinson's disease (or related disorder) followed up in the Parkinson Expert Centers or sites members of the NS-PARK network.
Individuals at risk of developing Parkinson's disease (or related disorder) may also be included in the cohort. These are i) people related to a PD patient, or ii) with a genetic risk factor for the disease, or iii) with a prodromal form1 of the disease, according to the current diagnostic criteria.
During a follow-up consultation, the neurologist of the site participating in the project will present the objectives and the modalities of participation in the cohort to the patient (Information & Opposition), orally and with a written information notice. If the patient wishes to participate, his express consent is collected by the physician.
Access to research results
A dedicated section (Publications), which is regularly updated, provides access to the global results of research conducted on the NS-PARK cohort.
Within this cohort, clinical, biological and imaging data are collected longitudinally:
- Demographic data (e.g. age)
- Clinical data related to Parkinson's disease
- Biological data from optional or existing samples (care or other research projects)
- Imaging data from optional or existing imaging exams (care or other research projects)
The researchers working on the study will link all these data during analyses aiming to better understand Parkinson's disease and to indentify subgroups of patients sharing common progression profiles of the disease, in order to eventually propose targeted therapies.
These analyses will thus allow to answer:
- To the specific objectives of the NS-PARK cohort
- To associated objectives; researchers working on Parkinson's disease can indeed conduct data analysis projects on the NS-PARK cohort, in order to answer scientific questions on the same theme. These studies must be validated by the Scientific Committee of the cohort and have the necessary regulatory authorizations.
Cohort tools
In order to implement the collection of data and samples for the cohort, various technical tools are used by the research teams. Only authorized study staff have access to the data and samples collected.
Online data entry database
Data collected during the inclusion in the cohort and follow-up visits are recorded by research professionals through secure access to an electronic case repart form (REDCapTM eCRF*; https://www.project-redcap.org). All participating centers have the same interface, for a standardized and homogenous data collection. Data extraction from the system, for analysis, is carried out in accordance with regulatory requirements in terms of confidentiality.
Biological Resource Center
Biological samples collected as part of the cohort are sent under coded labeling to the DNA & Cell Bank in the Brain and Spine Institute (Institut du Cerveau et de la Moelle - ICM) at the Pitié Salpêtrière Hospital, in Paris (https://institutducerveau-icm.org/en/biological-resource-center/). This centralization will allow standardized storage and processing of samples in a certified quality environment (AFNOR NF S 96900).
Multicenter neuroimaging platform
The brain images collected in the cohort are centralized at the Centre d’Acquisition et de Traitement d’Images (CATI, https://cati-neuroimaging.com) in Paris, which is responsible for the harmonization (monitoring, standardization) of imaging data of the project.
*An electronic Case Report Form is used to collect online data useful for the research
Organization of the cohort
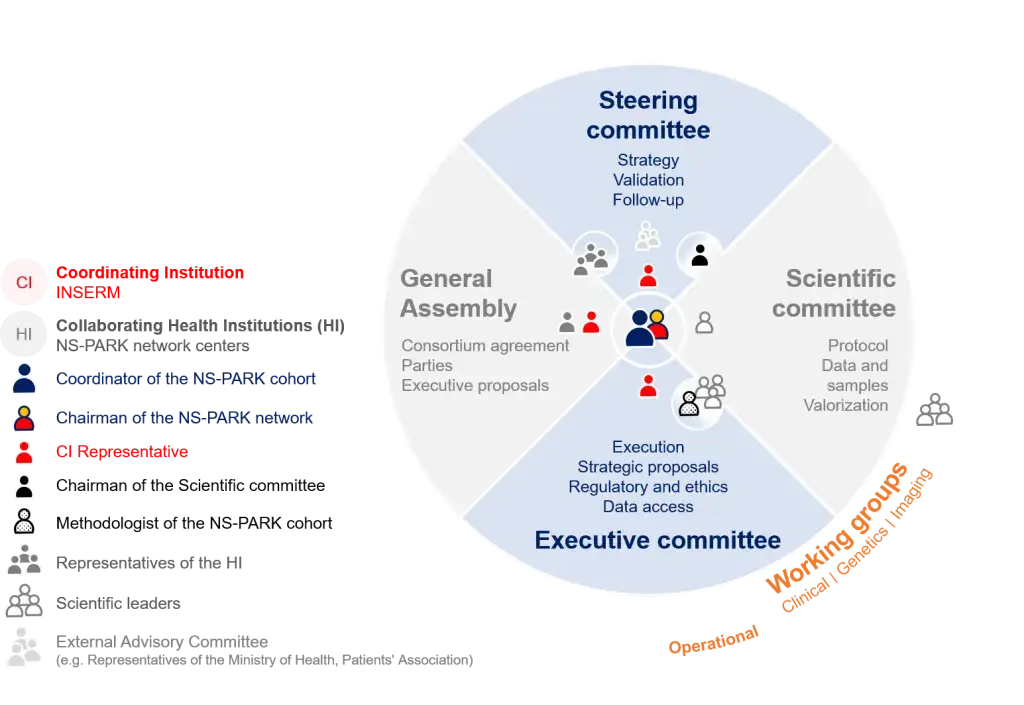
List of contributors
| Coordinating Institution (CI) |
INSERM Institut National de la Santé et de la Recherche Médicale (National Insitute of Health and Medical Research) |
| Collaborating Health Institutions (HI) | NS-PARK network centers |
| Representatives of the HI of the Steering Committee | University hospitals of Bordeaux, Lille and Lyon |
| CI Representative | Dr Hélène Espérou, Director of the Inserm Clinical Research Unit |
| Coordinator of the NS-PARK cohort | Prof. Jean-Christophe Corvol |
| Chairman of the NS-PARK network | Prof. Olivier Rascol |
| Chairman of the Scientific committee | Prof. Franck Durif |
| Methodologist of the NS-PARK cohort | Prof. Florence Tubach |
| Scientific leaders of the Executive committee | Prof. Luc Defebvre, Prof. Wassilios Meissner, Prof. Stephane Thobois |
| Scientific leaders of the HI | Scientific leaders of the NS-PARK network centers |
Data collected in the cohort
Implementation
The participating centers have a minimum data sheet to fill in the online database at each patient visit. This "Parkinson data sheet", whose items have been defined by the NS-PARK network experts, gathers essential data for patients follow-up :
- Demographic and activity data (e.g. age, gender, type and date of visit)
- Diagnosis (e.g. selected diagnosis, date of diagnosis)
- Clinical data : items of the MDS-UPDRS scale ; motor symptoms (e.g. motor fluctuations, dyskinesia, freezing) and non motor symptoms (e.g. hypotension, constipation problems, urinary problems, sleep disroder, pain, cognitive impairment, psychiatric disorder)
- Current antiparkinsonien treatment (type, class, daily dose)
Only authorized study staff have access to the collected data.
Analysis
The progression of the severity stage (Hoehn and Yahr stage), the onset and evolution of motor and non-motor symptoms of the disease will be evaluated in the whole population of patients included in the cohort, taking into account the chosen therapeutic strategy.
Implementation
Optionnal biological samples are proposed to participants included in the cohort (see the table below).
Biological residuals* from care or other research projects can also be collected for analysis in the NS-PARK cohort.
The collected samples are transferred to the Biological Resource Center of the ICM (Paris). Only authorized staff have access to the collected samples. The analyses planned on these samples are described in the table below.
Analysis

*The biological residual is the remaining volume of a biological sample originally collected as part of the patient's routine follow-up or from another study in which the patient participated.
Implementation
Existing imaging exams (DaTSCAN, MRI, PET SCAN, 18F-FDOPA PET), from care or other research projects, can be collected for analysis in the NS-PARK cohort.
An optional Magnetic Resonance Imaging (MRI) project specifically for the cohort has also been approved to collect specific imaging data, defined by a specialized working group of the NS-PARK network (NS-PARK MRI acquisition sequences). NS-PARK-Imaging will start in 2023 !
The collected images are transmitted to the CATI (Center for Acquisition & Image Processing, Paris, France) for harmonization and storage. Only authorized study staff have access to the collected imaging data.
Analysis
The analysis of brain imaging data acquired in a subgroup of patients in the cohort aims to identify imaging markers associated with the disease progression.

